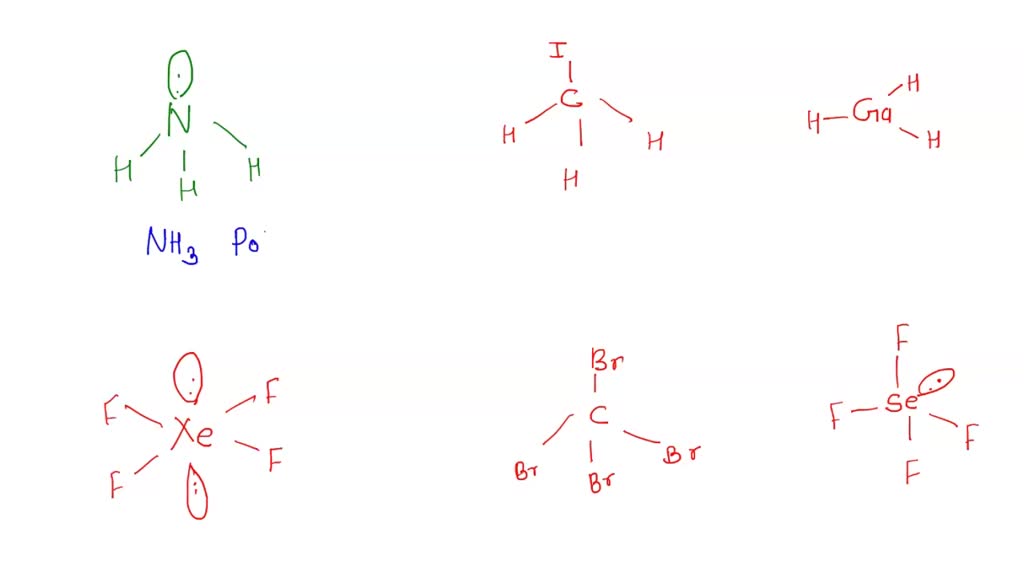
Chemistry 1110 Covalent Properties I. Assign electronegativity values to the following elements: N I S P H B O F Br C II. Tell

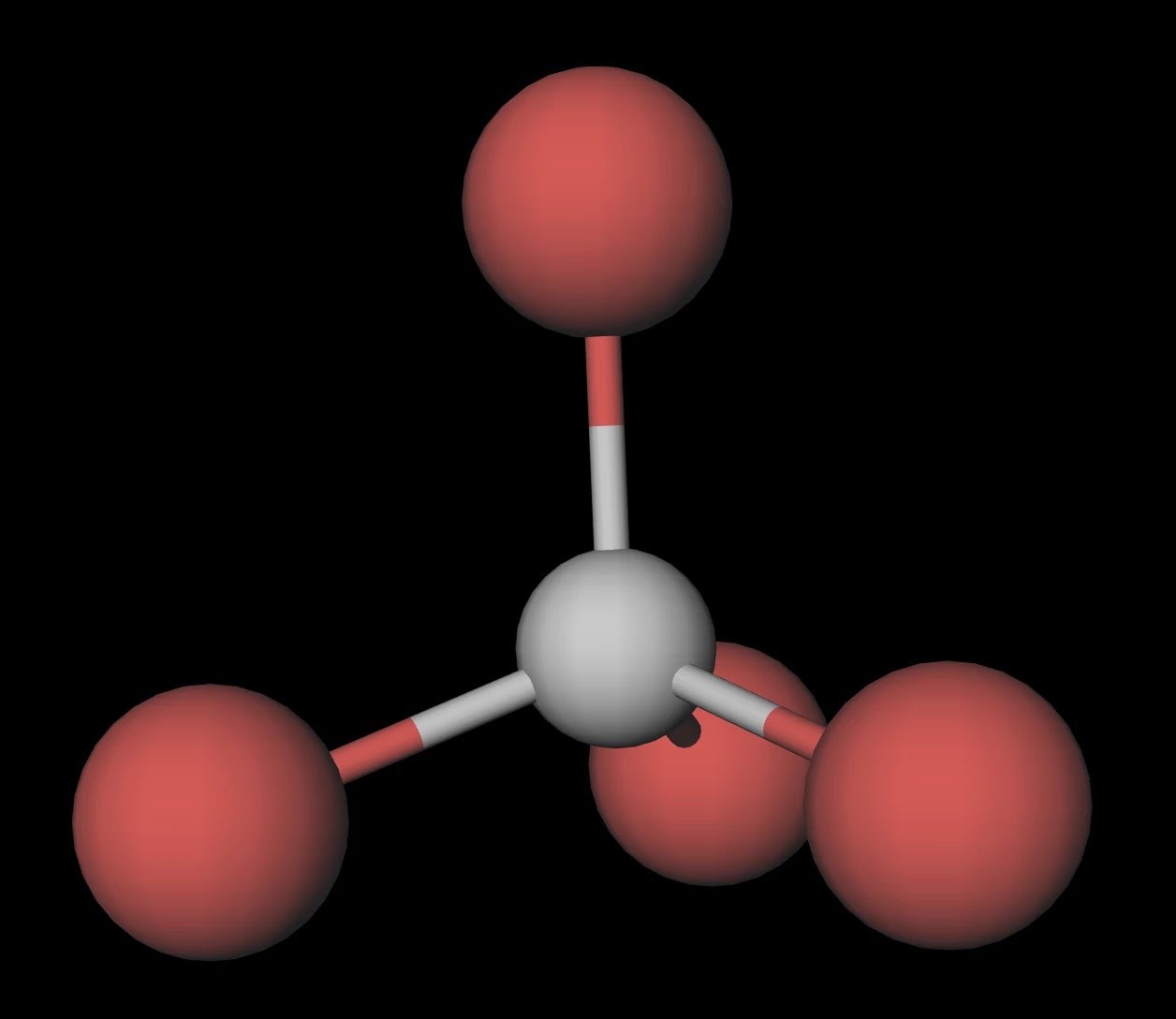
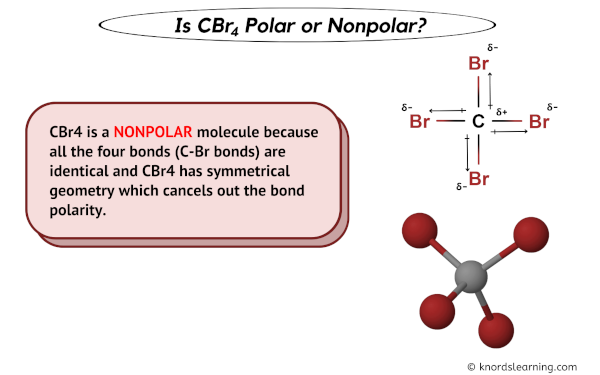
Draw the Lewis structure for CBr4 and provide the following information. a. number of electron groups b. electron pair geometry c. bond angle d. number of bonded electrons e. molecular geometry f.