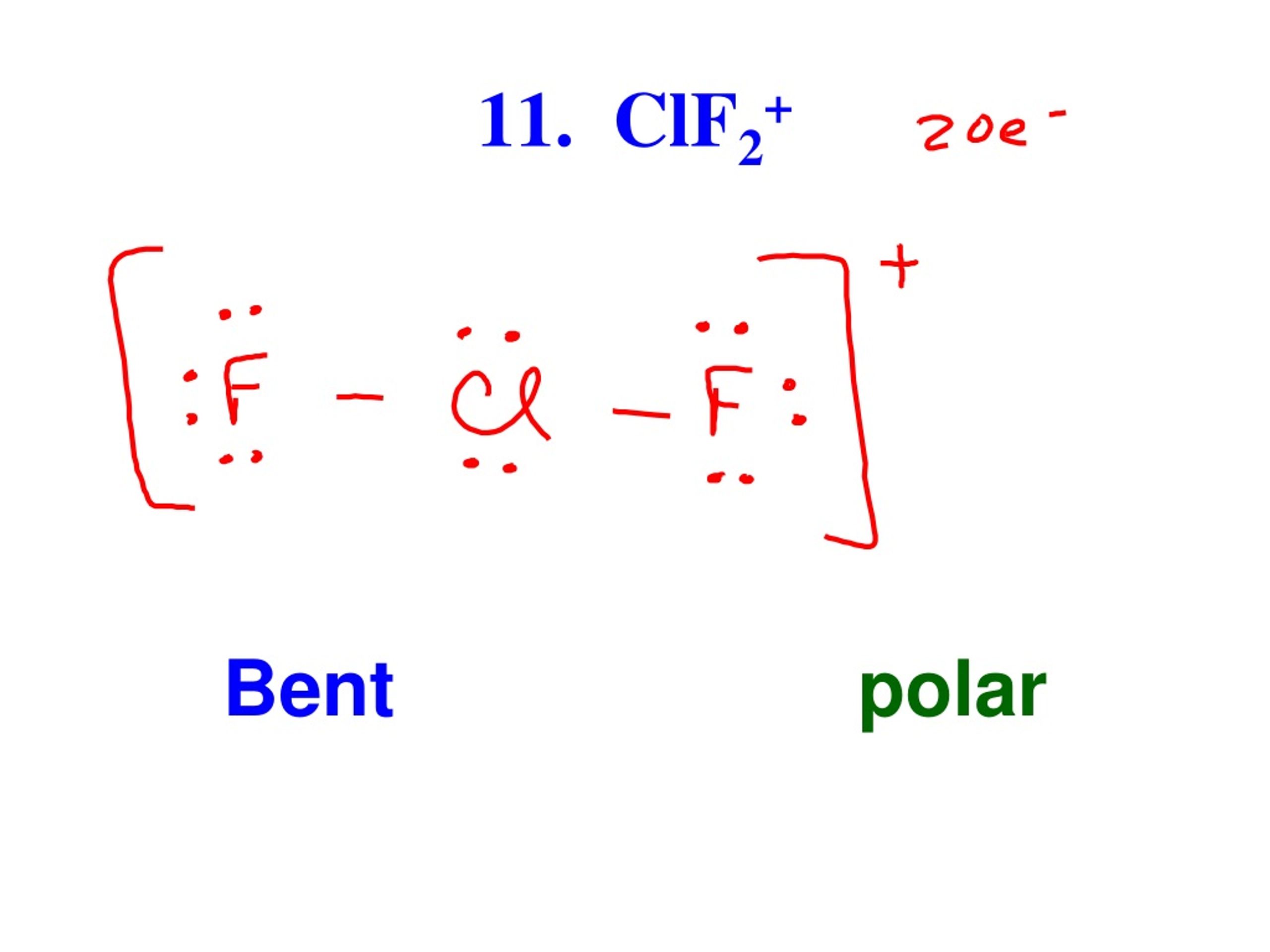
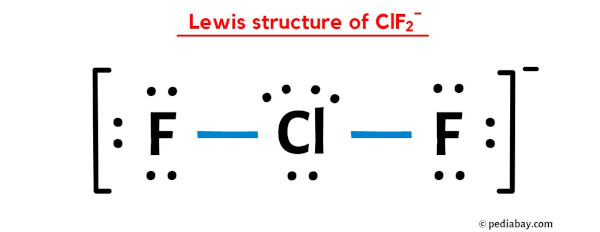
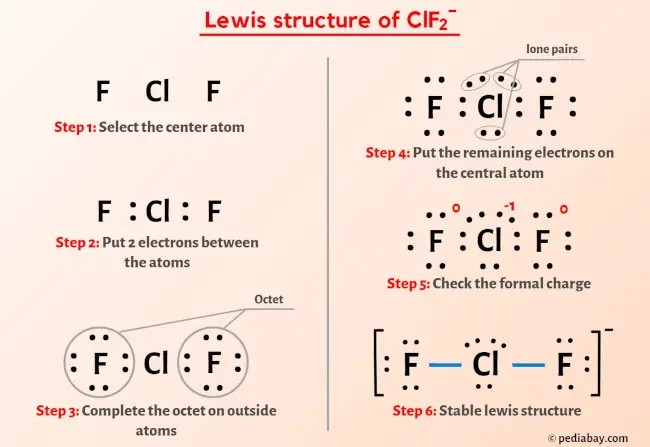
clf2-is linear while clf2+ is v shape why - Chemistry - The p-Block Elements - 10649819 | Meritnation.com

Your Turn! A central atom has two lone pair of electrons around it and two single bonds to other atoms. What is the electron pair geometry around the central. - ppt download

_____Good evening _____Answer this:-Explain why CIF2- is linear but CIF2+ is bent molecular ion. - Brainly.in

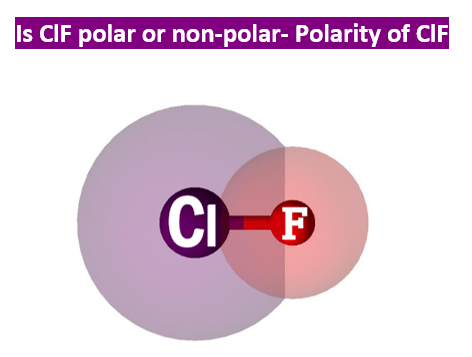
Is ClF Polar or Nonpolar (Chlorine Monoluoride) | Is ClF Polar or Nonpolar (Chlorine Monoluoride) Hello Everyone! Welcome back to our channel, and for today's video, we will help you determine if...
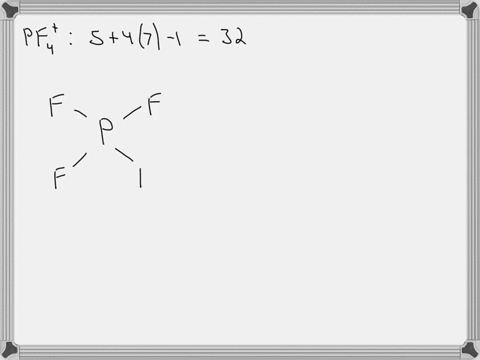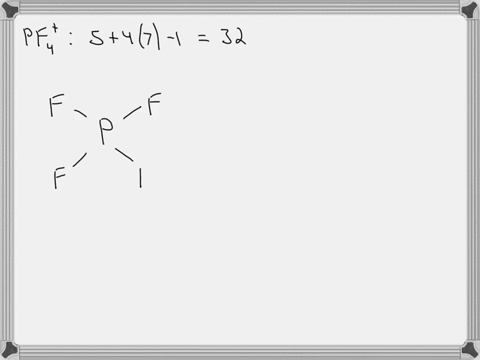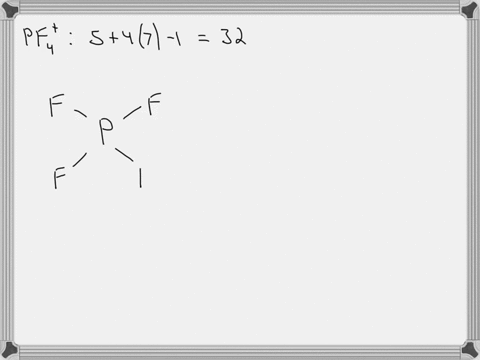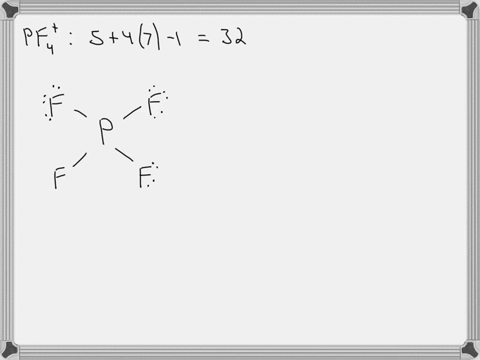
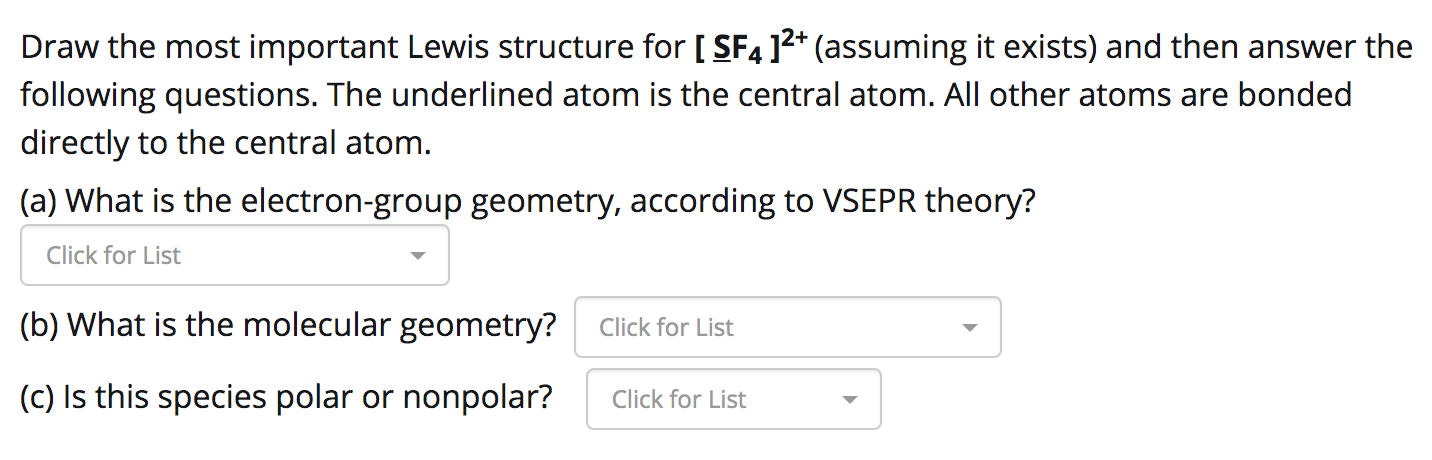
SOLVED: Answer the following for the indicated molecule or ion: Draw the complete Lewis structure of the molecule or ion. Determine the total number of electron groups (clouds) surrounding the central atom.

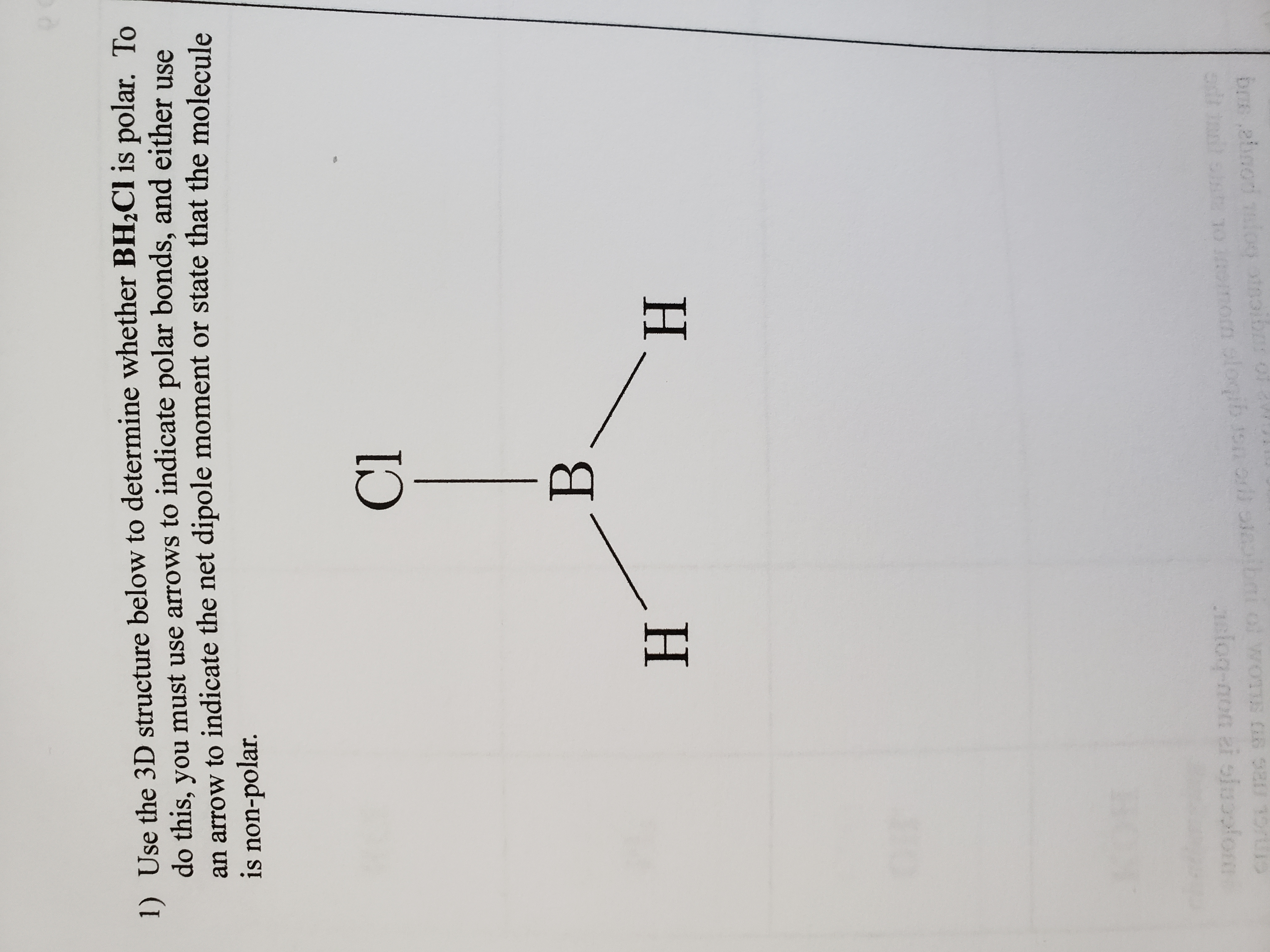
These are examples of molecules and ions that do not obey the octet rule. After drawing the Lewis structure of each, describe the electron region geometry and molecular geometry for each. a.

PS 15 key - Problem Set 15 – Molecular Shape, VSEPR Chem 105 1. Once you know the concepts of bond - Studocu