![What is the bond polarity of \\[N{H_3}\\]?A) \\[N{H_3}\\] is a non-polar moleculeB) \\[N{H_3}\\] is a polar molecularC) \\[N - H\\] bond is polarD) Both B and C What is the bond polarity of \\[N{H_3}\\]?A) \\[N{H_3}\\] is a non-polar moleculeB) \\[N{H_3}\\] is a polar molecularC) \\[N - H\\] bond is polarD) Both B and C](https://www.vedantu.com/question-sets/0f8d7d3e-59fc-4e46-bb6f-9eeb5b3d71541941507909701512327.png)
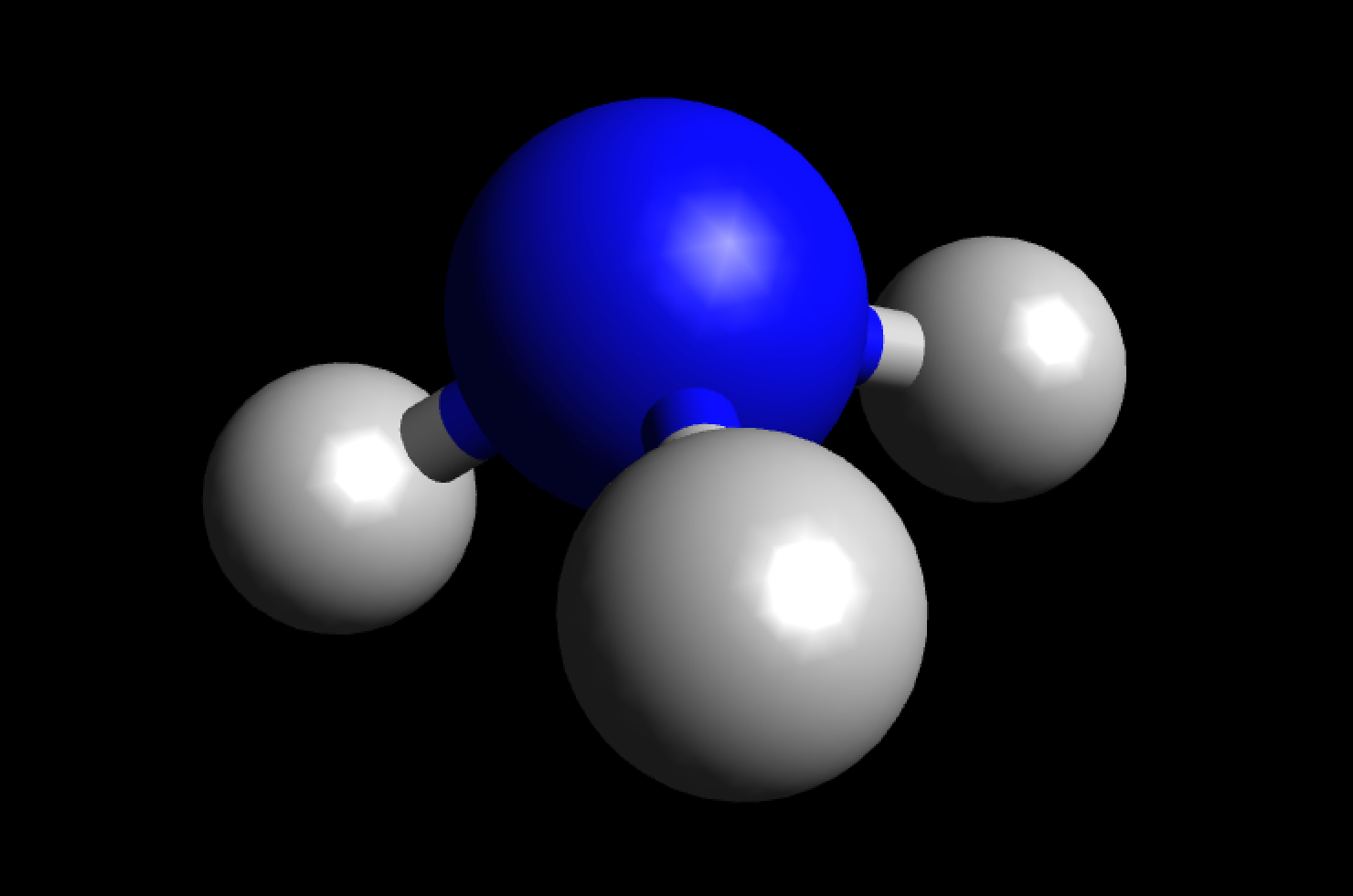
What is the bond polarity of \\[N{H_3}\\]?A) \\[N{H_3}\\] is a non-polar moleculeB) \\[N{H_3}\\] is a polar molecularC) \\[N - H\\] bond is polarD) Both B and C
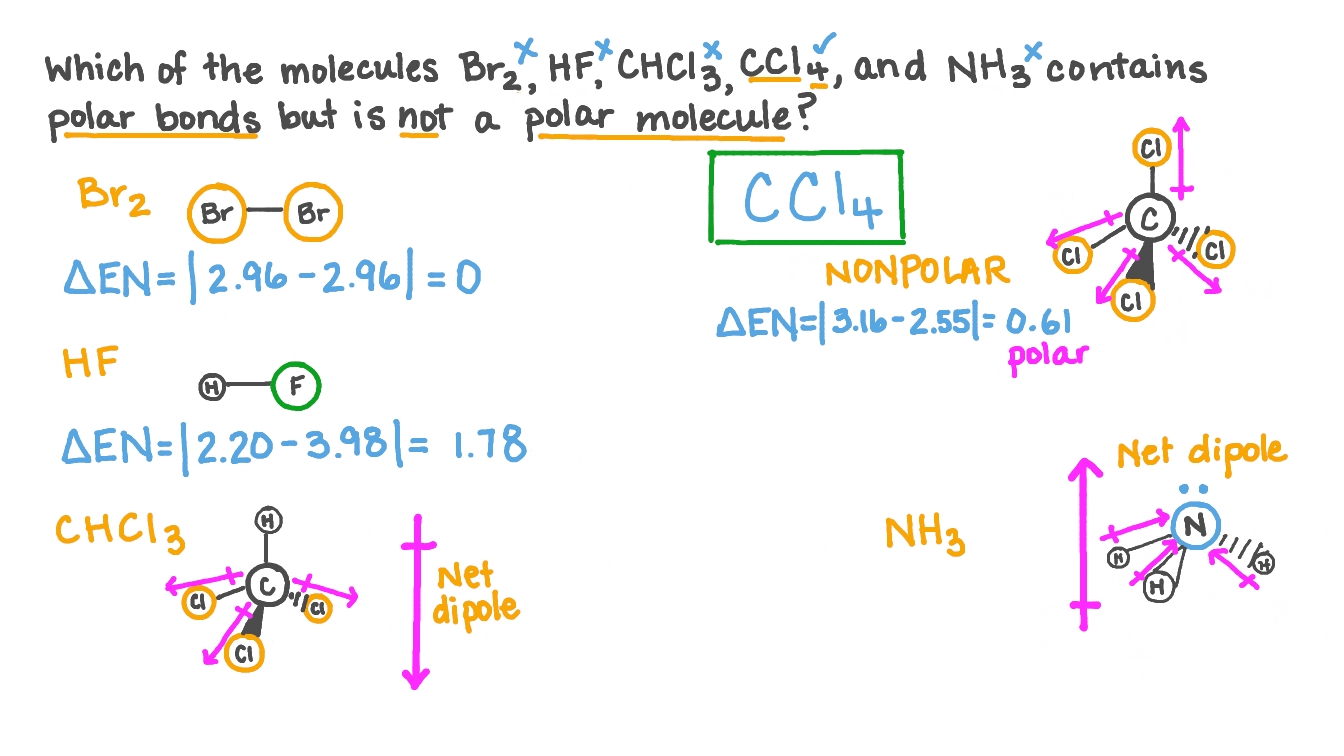
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa


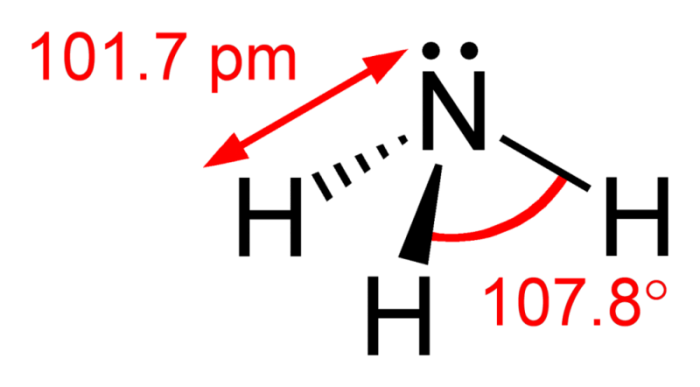
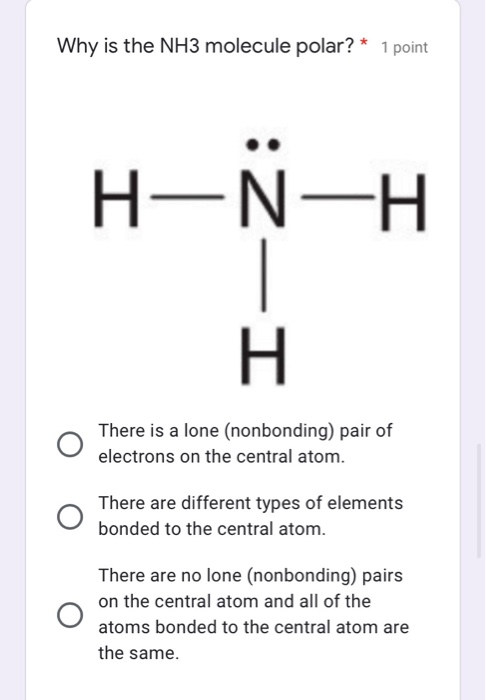





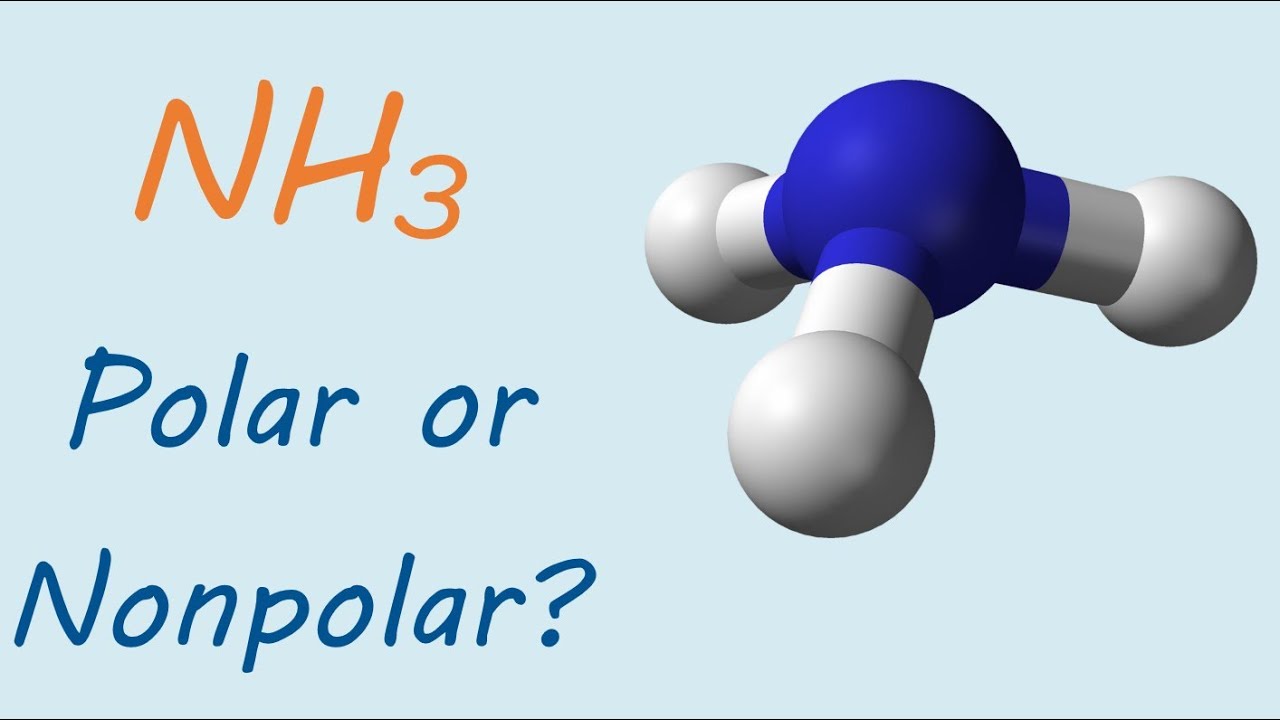

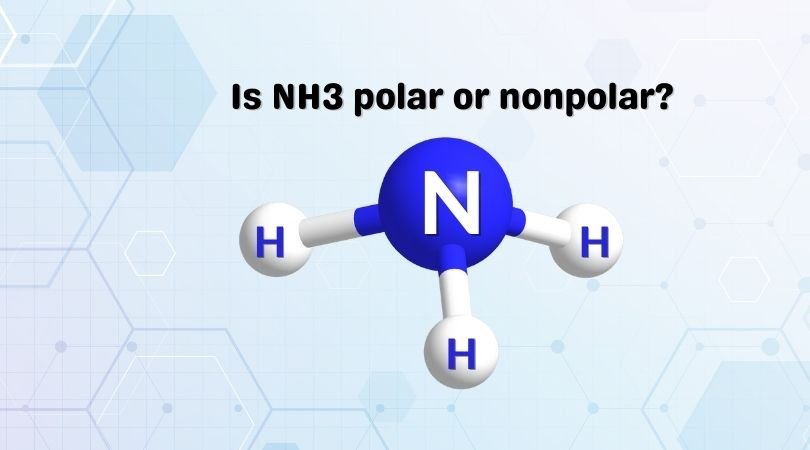
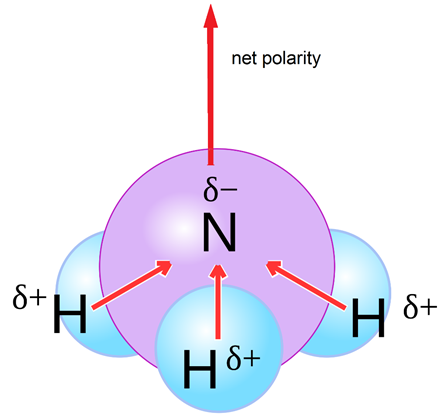
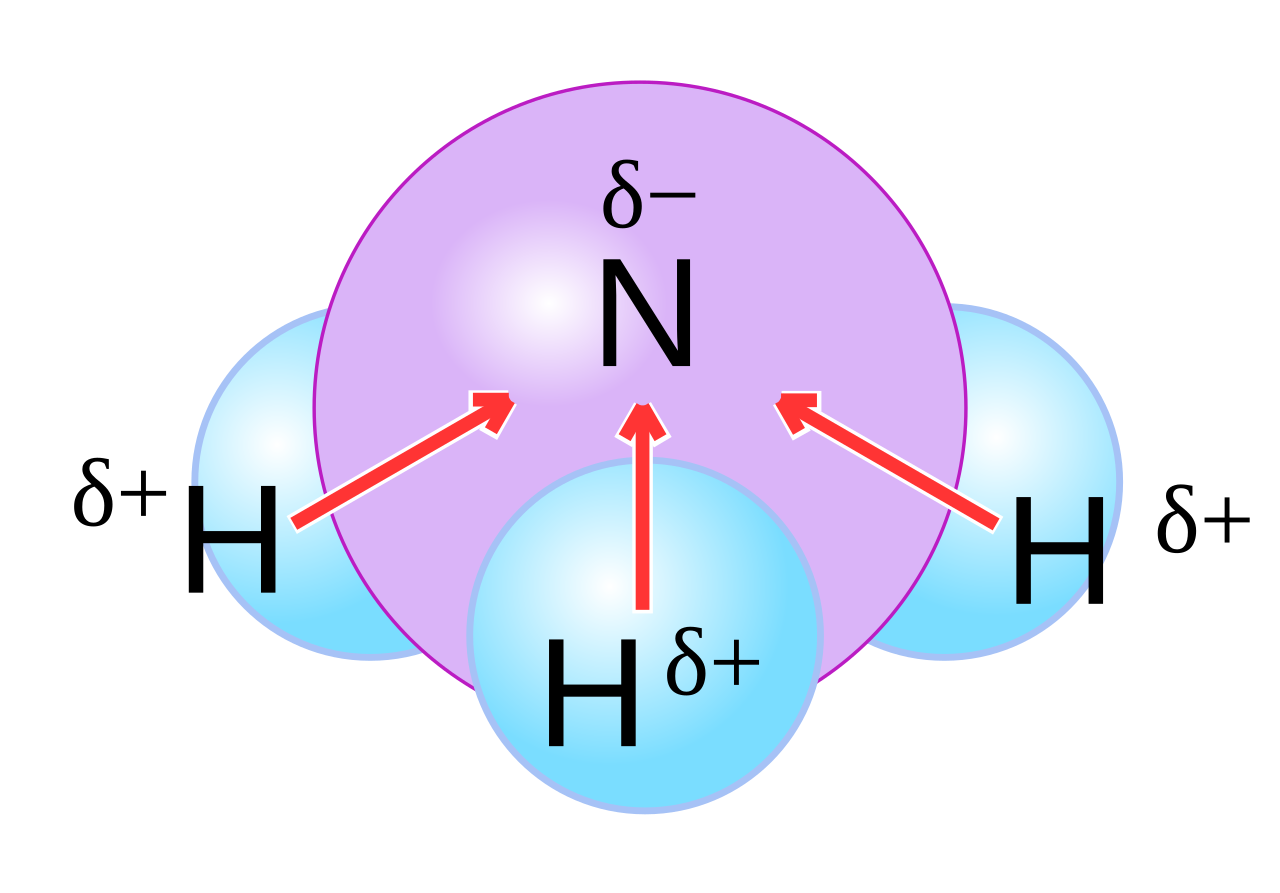


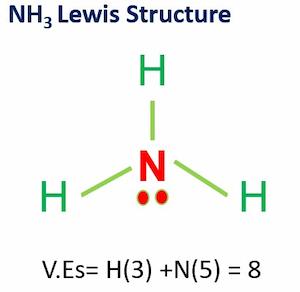



![Is NH3 Polar or Nonpolar [#1 Best explanation] - Science Education and Tutorials Is NH3 Polar or Nonpolar [#1 Best explanation] - Science Education and Tutorials](http://sciedutut.com/wp-content/uploads/2021/05/Is-NH3-Polar-or-Polar.png)