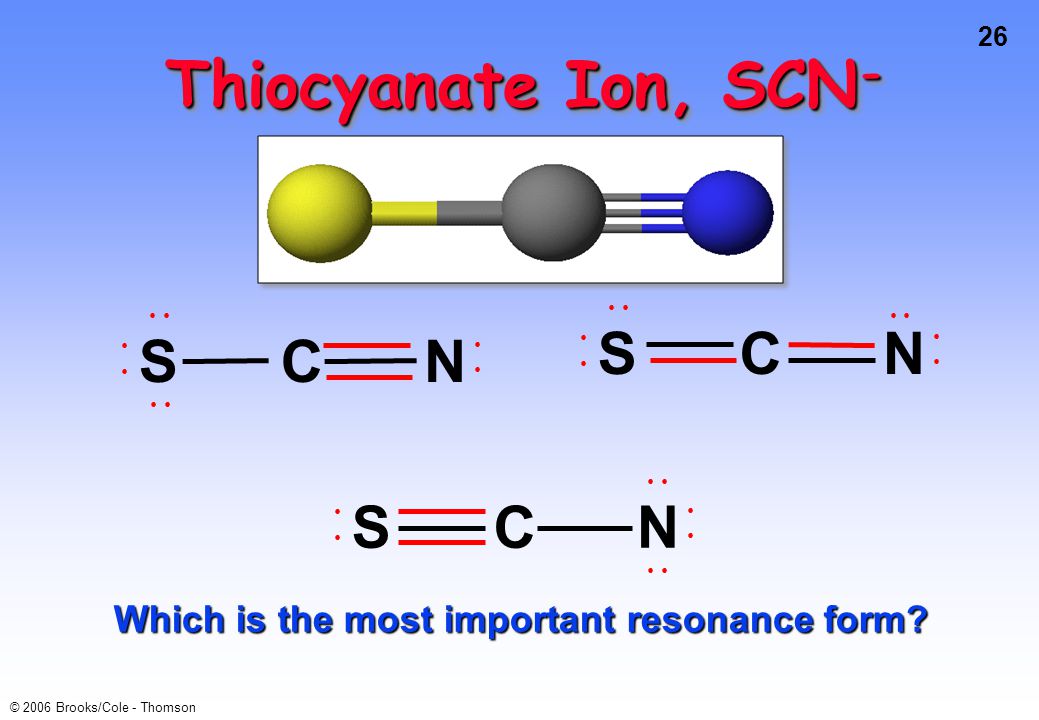
SOLVED: Decide whether each molecule or polyatomic ion is polar nonpolar. the molecule or polyatomic on is polar, write the chemical symbol of the atom closest to the negative side For example;

SOLVED: molecule or polyatomic ion polar or atom closest to nonpolar? negative side polar nonpolar polar nonpolar polar nonpolar Br SCN NH,

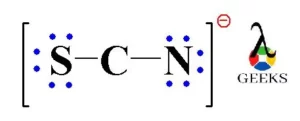
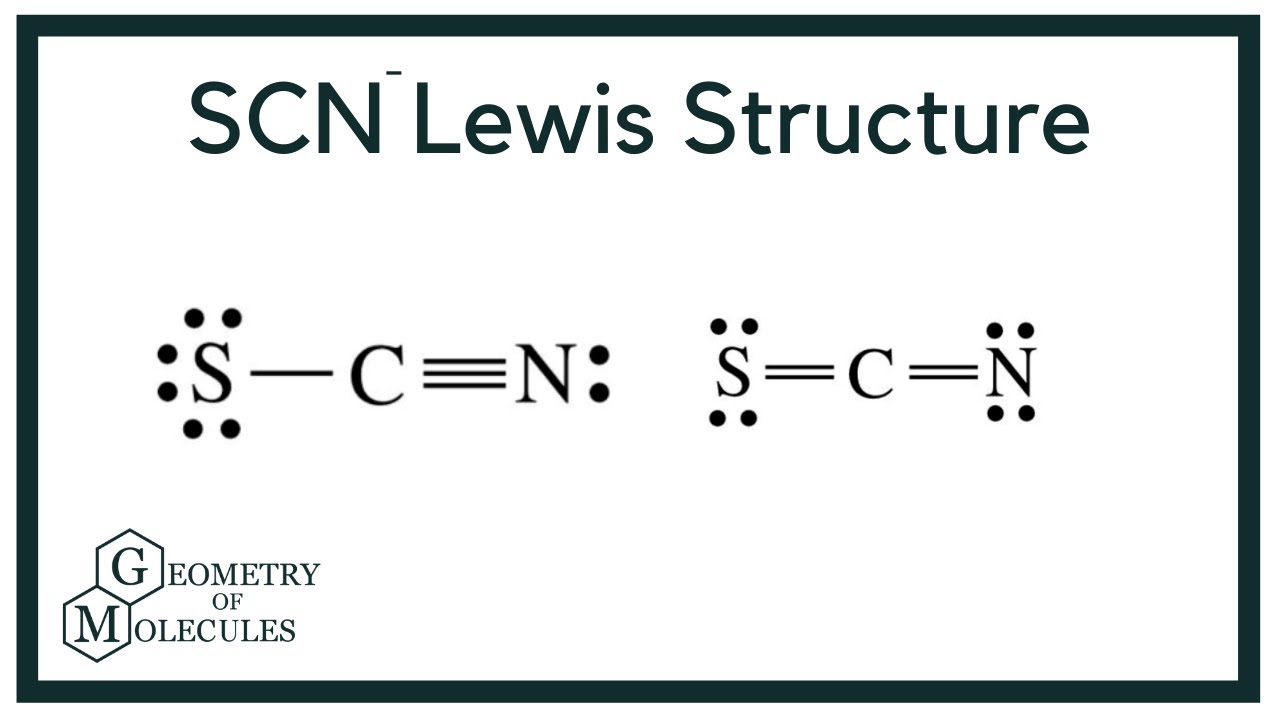
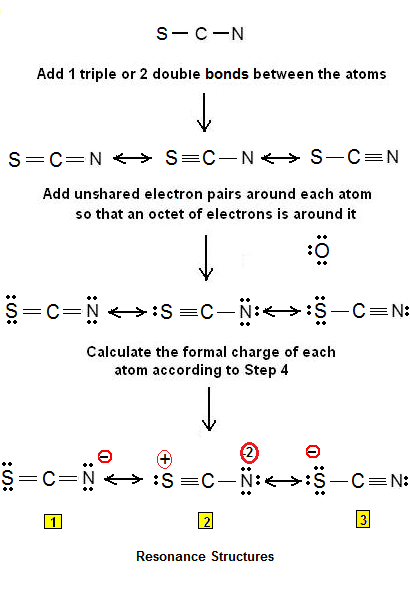
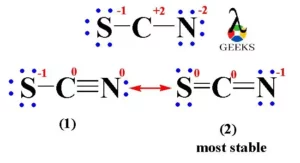
Draw the Lewis structure for SCN- and state its molecular geometry. Is it polar or nonpolar? | Homework.Study.com